Solutions
Law firms
Tax, audit & accounting firms
Success stories
-
A true competitive advantage
Law firm Zarwin Baum’s embrace of generative AI as the natural next step in the evolution of legal work and their adoption of CoCounsel Legal has helped them achieve remarkable efficiency gains and improved client relationships.
-
Workflow transformation drives impact
Brinks, a global leader in secure logistics and security solutions, used CoCounsel to reimagine what was possible with AI tools, turning legal challenges into a competitive advantage.
-
The forefront of audit tech
A better auditing workflow solution was the answer to multiple challenges faced by The Mercadien Group. Find out how they achieved greater efficiency by embracing Cloud Audit Suite.
Products
Legal
Trade & supply
Tax, audit & accounting
- 1040SCAN
- Audit Intelligence Analyze
- CoCounsel Audit
- CoCounsel Tax
- Ready to Advise
- Ready to Review
- View all
Corporate tax
Recommended products
-
CoCounsel Legal
Transform your work with the only AI legal solution uniting research, drafting, and document analysis in a single experience. Designed by legal experts and built on trusted content and advanced AI, CoCounsel Legal accelerates multistep work so you can better serve your clients and grow your business.
-
CoCounsel Tax
Transform your tax practice with CoCounsel Tax, an AI-powered assistant that combines trustworthy answers, automation, and firm knowledge into one seamless platform. Enhance efficiency, reduce risk, and improve client confidence with CoCounsel Tax.
-
CLEAR Investigate
Intelligently surface critical connections and insights in actionable format through AI-driven research workflows seamlessly integrated with the trusted and transparent CLEAR platform. Accelerate investigations and empower your team to make strategic decisions with confidence.
Purchase
Buy solutions
Resources
Insights
Events
Product training
Product communities
Developers
Highlights
-
2026 SKILLS showcase
Join weekly sessions to experience in-depth demonstrations of the leading legal AI products while connecting with strategic law firm leaders in knowledge management, innovation, and AI.
-
Ghosts on the ledger
Payroll fraud is a major compliance risk. Learn how payroll analytics and AI-powered tools can help exorcise phantom employees and employers.
-
Future of professionals report 2025
The Thomson Reuters Future of Professionals Report 2025 reveals how AI continues to shape professional work — and what it takes to get ahead. This year’s report shows that increased efficiency, productivity, and cost savings top the list of benefits professionals attribute to AI, making it indispensable for organizations facing rapid change.
Affirmation [PG23]
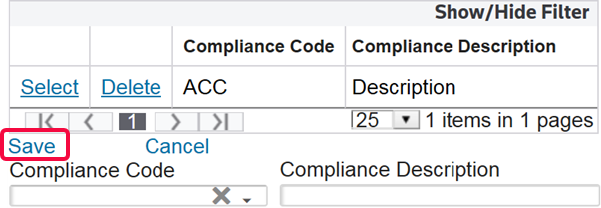
Links | Description |
|---|---|
Save | Save any data that was added or changed since the most recent save in Edit Mode. |
Cancel | Discard all changes made after the most recent save. The tab will revert to Read Only Mode. |
Edit | Activate Edit Mode so any necessary additions/modifications can be made to the open record. This action will only be made available once one or more records exist on the tab. |
Add New | Add a new record. This action will only be made available once one or more records exist on the tab. |
Delete | Delete the selected record. This action will only be made available once one or more records exist on the tab. |
- Enter the appropriate information in the fields listed below.
- ClickSave.
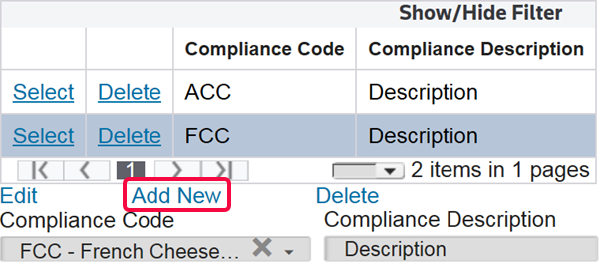
Columns | Description |
|---|---|
Compliance Code | A code used to affirm compliance with FDA requirements. If the merchandise is subject to the Bioterrorism Act (BTA), use this field to report the appropriate affirmation of compliance information not reported elsewhere. For a list of valid codes, refer to Appendix PGA (Food & Drug Affirmation of Compliance Codes). |
Compliance Description | Text describing the information required by the PGA. This could include a number or a country code, etc. For a list of valid codes related to certain specific Affirmation of Compliance Codes, refer to Appendix PGA (Food & Drug Affirmation of Compliance Qualifier Codes). |
This article applies to:
- Product: ONESOURCE Global Trade
- Sub-Product: US Regimes & Connectivity
Related Content
-
PGA Data Screen (PGA Mapping)
Format:Article, 1 min read
